When you work in a lab or handle chemicals, waste isn’t just trash – it’s a responsibility. One of the first things you’ll notice when sorting chemical waste is that it’s often labeled as either halogenated or non-halogenated. The difference might sound technical, but it’s actually pretty simple and important. Non-halogenated waste refers to chemical waste that doesn’t contain halogen elements like chlorine, bromine, or fluorine. Understanding this distinction helps keep disposal safe, compliant, and environmentally sound.
Understanding Non-Halogenated Waste
If you’ve ever worked in a lab, you’ve probably seen waste containers labeled “halogenated” and “non-halogenated.” It might look like a small detail, but it’s one that really matters. Non-halogenated waste refers to chemical waste that doesn’t contain halogen elements such as chlorine, fluorine, bromine, or iodine. Those elements are what make some solvents more toxic and harder to dispose of safely. By separating them out, we prevent dangerous chemical reactions and make the disposal process cleaner and easier to manage.
In most labs, non-halogenated waste includes familiar solvents like ethanol, acetone, methanol, ethyl acetate, and toluene. These are used all the time in cleaning and experiments, so the waste adds up quickly. Even though these chemicals are considered less hazardous than halogenated ones, they can still cause fires, release strong fumes, and pose environmental risks if handled carelessly.
Properly managing this waste helps protect people working in the lab as well as the environment around us. It ensures that waste can be safely recycled or burned for energy without producing harmful byproducts. Paying attention to how we separate and store non-halogenated waste might not seem like a big deal, but it’s one of the simplest ways to keep a lab safe, compliant, and responsible.
Why the Difference Matters
The difference between halogenated and non-halogenated waste is more than just a chemistry term. It determines how waste is collected, stored, and disposed of. Halogenated solvents, which contain elements like chlorine or bromine, can release highly toxic gases when burned. They also tend to linger in the environment longer and require specialized treatment. Non-halogenated solvents, on the other hand, can usually be burned more safely for energy recovery or recycled through solvent distillation.
Even a small amount of mixing between the two can cause problems. Once a halogenated solvent enters a container of non-halogenated waste, the entire batch must be treated as hazardous halogenated waste. That not only increases disposal costs but also adds more environmental risk. For this reason, Environmental Health and Safety (EHS) teams emphasize strict separation and labeling practices in every lab.
From a compliance perspective, keeping these waste streams separate is part of meeting the EPA’s Resource Conservation and Recovery Act (RCRA) requirements. Clear segregation helps institutions stay compliant, avoid fines, and maintain cleaner, safer labs. Beyond regulations, it’s simply a good habit that protects people, budgets, and the planet.
Common Examples of Non-Halogenated Waste
Non-halogenated waste comes from everyday lab activities such as cleaning, chemical synthesis, and solvent extraction. Although specific chemicals vary depending on the type of research, most fall into a few common categories.
Alcohols
Alcohol-based solvents are widely used for cleaning, reactions, and sample preparation.
- Ethanol
- Methanol
- Isopropanol
Ketones
Ketones are strong solvents that evaporate quickly, making them useful for cleaning and dissolving resins or coatings.
- Acetone
- Methyl ethyl ketone (MEK)
Ethers
These are common in extractions and reactions but must be handled carefully due to flammability and peroxide formation.
- Diethyl ether
- Tetrahydrofuran (THF)
Aromatic Solvents
Aromatic hydrocarbons are often used for degreasing, dissolving nonpolar compounds, and in some analytical procedures.
- Toluene
- Xylene
Esters
Esters are found in cleaning agents and as reaction solvents.
- Ethyl acetate
- Butyl acetate
Key Properties and Handling Notes
- Flammability: Most non-halogenated solvents have low flash points, so they must be kept away from heat or open flames.
- Volatility: Many evaporate quickly, producing vapors that can cause irritation or dizziness in poorly ventilated areas.
- Reactivity: While generally stable, they can react dangerously with oxidizers or acids if stored incorrectly.
- Environmental Impact: When handled properly, these wastes can often be recycled or incinerated for energy recovery without generating harmful halogenated byproducts.
Why Proper Identification Is So Important
Mislabeling or mixing non-halogenated waste with halogenated waste doesn’t just cause confusion–it completely changes how it must be treated. A single drop of chloroform, for instance, can contaminate a whole container of acetone, forcing disposal as halogenated waste. That means higher costs, stricter handling, and additional environmental burden.
By clearly labeling containers and keeping halogenated solvents separate, labs can reduce risk, cut costs, and stay compliant with environmental standards. It’s one of those small tasks that has a big impact on safety and sustainability.
How Non-Halogenated Waste Is Classified
Even though non-halogenated waste doesn’t contain halogen elements, it doesn’t automatically mean it’s harmless. Many of these materials still count as hazardous waste under the U.S. Environmental Protection Agency (EPA) standards. The EPA looks at the physical and chemical characteristics of each waste type to decide how it should be managed.
EPA Criteria for Hazardous Waste
The EPA classifies a waste as hazardous if it meets one or more of the following traits:
Ignitability
Waste is considered ignitable if it can catch fire easily.
- Has a flash point below 60°C (140°F)
- Common examples include acetone, ethanol, and toluene
- Must be stored away from open flames or heat sources
Corrosivity
Corrosive waste can wear down metal or damage containers.
- Usually has a pH lower than 2 or higher than 12.5
- Can damage surfaces and equipment
- Should be stored in containers that resist corrosion, like plastic or stainless steel
Reactivity
Reactive wastes are unstable under normal conditions.
- Can react violently with water or other substances
- May release toxic gases or even explode if mixed improperly
- Must be kept separate from oxidizers, acids, and other reactive materials
Toxicity
This classification is based on how harmful the waste can be to human health and the environment.
- Toxic wastes can leach contaminants into soil and groundwater
- Even small amounts can make a large volume of waste hazardous
- Testing and careful labeling are key to proper handling
The Takeaway
Non-halogenated waste may sound safer, but it often still falls under one or more of these categories. The goal is not to assume it’s risk-free but to recognize the potential hazards and handle it responsibly.
Storage and Labeling Guidelines
Proper storage and labeling of non-halogenated waste are at the core of safe lab operations. When these materials are managed the right way, it prevents accidents, ensures compliance, and simplifies the disposal process.
Approved Containers
Always collect non-halogenated solvent waste in approved containers, such as:
- High-density polyethylene (HDPE) bottles or jugs
- Metal containers rated for flammable liquids
- Containers with tight-fitting caps to minimize vapor release
Avoid using glass containers for large quantities, since they can break easily.
Labeling Requirements
Every waste container must have a clear label so there’s no confusion about its contents.
- Use pre-printed or EHS-approved labels marked “Non-Halogenated Waste.”
- Include chemical names and approximate percentages.
- Add hazard warnings like “Flammable” or “Toxic Vapors.”
- Write legibly using permanent ink, not pencil or erasable marker.
Proper labeling not only keeps lab workers safe but also helps EHS teams during waste collection and classification.
Storage Location and Conditions
How and where you store waste makes a big difference in preventing spills or fires.
- Keep containers closed at all times unless you’re adding or removing waste.
- Store in a flammable storage cabinet or designated accumulation area.
- Place containers away from ignition sources such as hot plates, Bunsen burners, or heat lamps.
- Use secondary containment trays to catch drips or leaks.
It’s also smart to keep halogenated and non-halogenated waste containers far apart, so there’s no risk of cross-contamination. Even a small amount of halogenated solvent can change how the entire batch must be disposed of.
Why Labeling and Storage Matter
It might feel like overkill to spend time labeling every bottle or checking storage locations, but it’s one of the most effective ways to prevent accidents. A mislabeled container could lead to dangerous reactions, while poor storage can increase fire risk or cause unnecessary exposure to fumes.
Most Environmental Health & Safety (EHS) departments have strict rules for a reason. Following them ensures safe pickup, compliant disposal, and peace of mind for everyone working in the lab. A few extra minutes spent labeling and sealing containers properly can prevent hours of cleanup and serious safety incidents later on.
Proper Disposal and Pick-Up Procedures
Once waste containers are full or near their limit, they should be scheduled for pickup through your institution’s EHS office. Disposal procedures can differ between organizations, but most follow a similar process:
- Submit a pickup request through an internal hazardous waste management system or form.
- Ensure the container is properly labeled and sealed.
- Include all constituents on the label, even if present in small amounts.
- Store containers temporarily in an approved accumulation area until collection.
Many institutions work with licensed waste management vendors that handle solvent recycling or energy recovery through incineration. Non-halogenated solvents, because they burn cleaner, can sometimes be used as fuel blends, which helps offset disposal costs and supports sustainability goals.
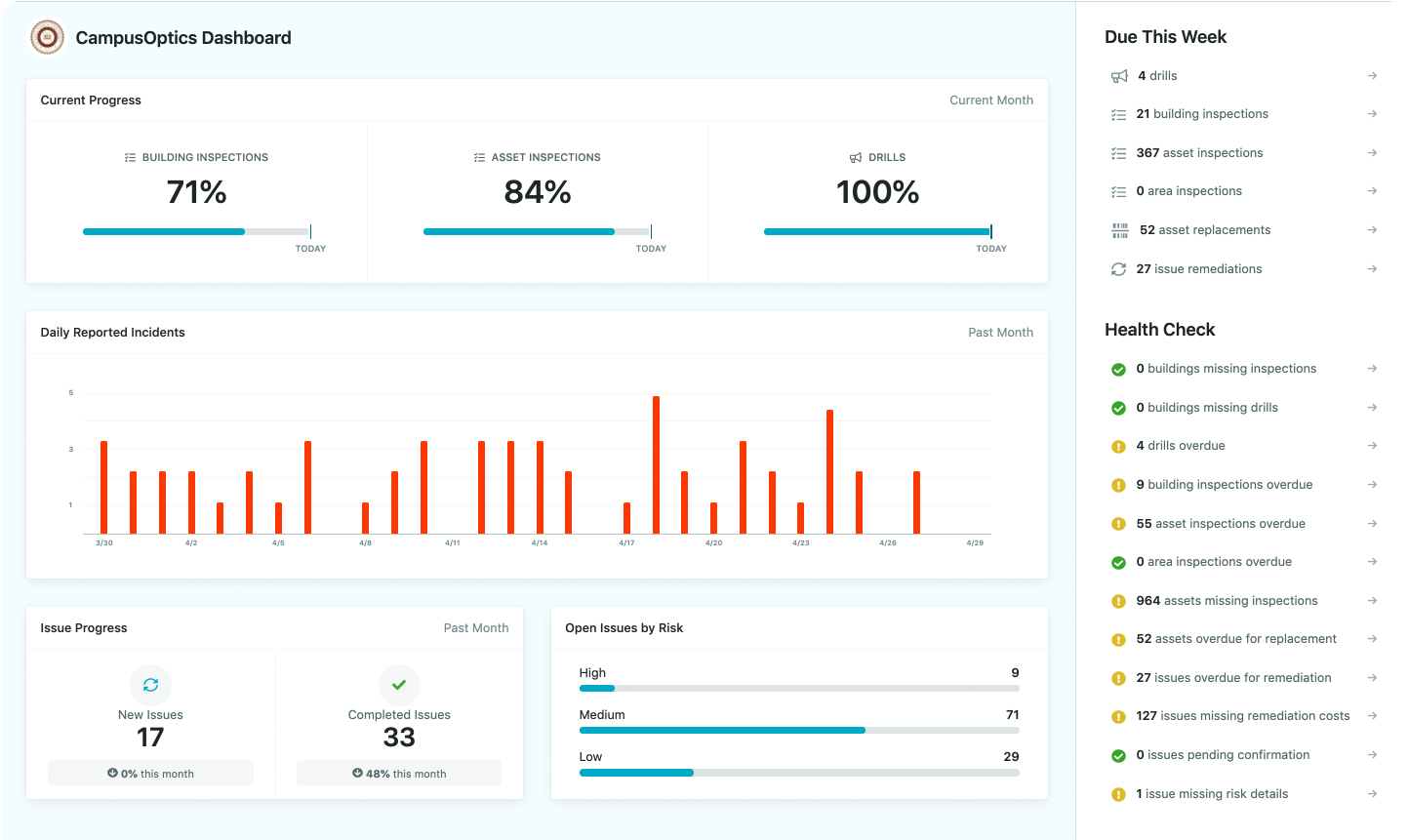
How We Support Safer Waste Management with CampusOptics
At CampusOptics, we see waste management as part of a bigger picture. Non-halogenated waste isn’t just something to dispose of correctly – it’s part of how a campus protects its people and the environment every day. Our platform helps colleges and universities manage every stage of that process, from chemical inventory to final waste pickup, in one connected system. We make it easier to track what’s in storage, monitor containers, and ensure that non-halogenated waste never gets mixed with halogenated materials or other incompatible chemicals.
Because campus safety professionals are rarely behind a desk, we built our tools to work wherever they are. Whether it’s scanning a container label, logging an issue during an inspection, or accessing Safety Data Sheets in the field, the information is always right at hand. We believe that keeping waste streams clean and compliant starts with visibility and collaboration – and that’s exactly what we aim to deliver through CampusOptics.
Why Mixing Waste Is a Serious Problem
It’s easy to assume that a few drops of leftover solvent won’t cause much trouble, but that small mistake can turn into a real issue. Mixing different types of chemical waste can change how it’s classified, how it reacts, and how it must be handled. Here’s why keeping them separate matters:
Contamination happens fast
Just a little bit of a halogenated solvent, like chloroform or dichloromethane, can contaminate an entire container of non-halogenated waste. Once that happens, the whole batch must be treated as halogenated, which means higher disposal costs and stricter handling rules.
The risk of dangerous reactions increases
Some chemicals simply don’t mix well. Combining oxidizers with organic solvents can cause violent reactions, fires, or even explosions. Keeping waste streams separate prevents these unexpected hazards.
It complicates disposal and tracking
When waste is mixed, it becomes harder for Environmental Health & Safety (EHS) teams to identify what’s inside and determine the safest disposal method. That leads to extra paperwork, delays, and possible compliance issues.
You lose recycling opportunities
Non-halogenated waste can sometimes be recycled or incinerated for energy recovery. Mixing it with halogenated solvents ruins that chance, increasing environmental impact.
Separating waste isn’t just a formality – it’s one of the easiest ways to protect people, save money, and stay compliant. A few seconds of caution when pouring into a container can prevent a chain of much bigger problems later on.
Environmental and Safety Considerations
Proper management of non-halogenated waste isn’t just about compliance. It’s also about reducing environmental impact and keeping people safe. Improper disposal can lead to soil and groundwater contamination or release of volatile organic compounds (VOCs) into the air.
Environmental Health and Safety (EHS) programs at universities, such as those at Temple University and Cornell, emphasize that every lab worker has a role in waste reduction. Simple actions, like limiting solvent use and keeping containers sealed, make a real difference.
Good waste management practices also protect the people working directly with chemicals. Breathing in solvent vapors or handling containers carelessly can cause dizziness, headaches, and skin irritation. Following safety protocols protects both the lab team and the environment.
Tips for Reducing Non-Halogenated Waste
Waste reduction starts with prevention. Here are a few practical ways to cut down on the amount of solvent waste your lab generates:
- Plan experiments carefully: prepare only as much solvent as you need.
- Use microscale techniques: smaller reactions mean less leftover material.
- Recycle and reuse solvents: when it’s safe and permitted by your institution.
- Substitute less hazardous materials: whenever possible.
- Regularly review inventory: to prevent chemicals from expiring or becoming unusable.
These small steps help build a culture of sustainability and safety in any research environment.
Training and Institutional Responsibility
Most academic and industrial organizations require anyone handling chemical waste to complete EHS waste management training. These sessions cover labeling rules, accumulation limits, emergency procedures, and contact points for reporting spills or container leaks.
Institutions like Cornell University outline clear expectations for their lab staff and students: segregate waste correctly, follow labeling guidelines, and never leave containers open. The Environmental Health & Radiation Safety (EHRS) office or equivalent department typically provides quick reference sheets and pickup schedules to make the process easier.
Creating accountability at every level, from lab technicians to principal investigators, helps ensure consistent compliance and reduces the risk of accidents or violations.
The Bigger Picture: Sustainability and Compliance
Proper management of non-halogenated waste is part of a broader effort to create sustainable lab environments. When labs correctly segregate waste, they make recycling and energy recovery more efficient. Clean solvent streams can be reused or repurposed, reducing both environmental footprint and disposal costs.
Regulatory compliance also plays a big role. The EPA, OSHA, and state environmental agencies all have regulations governing waste handling. Institutions that maintain clean, well-labeled waste streams show regulators that they take safety and sustainability seriously.
Ultimately, managing non-halogenated waste well reflects a culture of care – for people, the environment, and the research itself.
Conclusion
Understanding what non-halogenated waste is might not sound exciting at first, but it’s one of those behind-the-scenes things that keeps labs running safely and responsibly. These wastes are part of everyday research life – from acetone rinses to ethanol cleaning – and how we handle them says a lot about how seriously we take safety and sustainability.
The main idea is simple: even if these solvents don’t contain halogens, they still need care. Proper labeling, storage, and disposal prevent contamination, lower costs, and keep everyone in compliance with environmental standards. More than that, it builds a sense of accountability in the lab.
In the end, waste management isn’t just a rule to follow – it’s part of a bigger effort to protect people, research, and the environment. Paying attention to the small stuff, like separating halogenated from non-halogenated waste, is what allows science to keep moving forward without leaving a mess behind.
Frequently Asked Questions (FAQ)
1. What does non-halogenated waste actually mean?
It’s just a fancy way of saying chemical waste that doesn’t contain halogen elements like chlorine, bromine, fluorine, or iodine. In most labs, that means things like acetone, ethanol, methanol, and toluene – basically the everyday solvents you use for cleaning or reactions.
2. Why is it such a big deal to keep halogenated and non-halogenated waste separate?
Because mixing them can mess everything up. Even a small amount of a halogenated solvent can turn an entire container of non-halogenated waste into something much more hazardous. That means higher disposal costs and a lot more safety risks.
3. Is non-halogenated waste still considered hazardous?
Usually, yes. Even without halogens, most of these solvents are flammable, volatile, or toxic to some degree. So they’re still managed as hazardous waste under EPA rules – just a little less complex to handle than their halogenated counterparts.
4. How should I label a container for non-halogenated waste?
Keep it simple but clear. Write “Non-Halogenated Waste” on the label, list the main chemicals and their approximate amounts, and include any hazard warnings like “Flammable” or “Toxic Vapors.” Permanent marker, legible writing – nothing fancy, just accurate.
5. What’s the right way to store it?
Use an approved container, make sure it’s tightly sealed, and store it in a flammable cabinet or a designated waste area. Don’t leave lids open or keep containers near heat sources – it’s a small habit that prevents big problems later.